Chemistry of Anodization
General Information
1) The first thing to understand is that it has been determined through many experiments that Aluminum is an easy cheap and reliable way of making nanoporous membranes.
2) The second thing to understand is that the electrolyte used has virtually little to do with the anodization process. You will find that a lot of acids have been used and a few are favored for the results that are given but you can use just about any electrolyte. This of course excludes electrolytes that will corrode just about anything.
The Chemistry of Anodization
Now what I have gathered is that through the potential difference that is applied between the anode (Al foil usually only 0.25mm and always 99.99% pure) and the cathode (anything but I have been using a stainless steel plate) the 3 valence electrons on the Al metal are attracted so much by the positive anode that Al(III) forms and starts to drift off toward the negative cathode. These positive ions are immediately intercepted by O(-2) ions and even OH ions. By immediately I mean some nanometers off of the remaining Al metal.
Oxide layer: This is how the oxide layer starts to form but what ends up happening is that as one layer forms another layer forms on top of that. And then another and then another. Due to the properties of the electrolyte, the amount of voltage and the amount of current (which should be kept to no more than 0.5-1A otherwise the surface of the Al will be deformed) the oxide layer created straight pores (channels) that are nanometers wide. The uniformity of these pores are always in question and is difficult to control however not impossible. The pores that are formed are round/oval and have a concave end where the oxide layer meets the aluminum layer and for every one there are six other pores surrounding it giving a hexagonal symmetry. Now one of the questions I am still trying to understand is why these pores form a concave end instead of being flat and the surface of the aluminum and also exactly why these pores form at all.
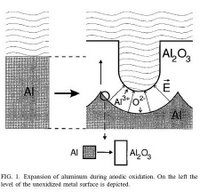
Reprinted with permission from: Self organized formation of hexagonal pore arrays in anodic alumina. O. Jessensky, F. Muller, U. Gosele., Applied Physics Letters, Vol 72 (10), 1173-1175, 1998
Copyright 1998, American Institute of Physics.
Reaction Equations:
[1] Al=Al(III) + 3e
[2] 2Al(III) + 3H2O = Al2O3 + 6H(+)
[3] 2Al + 3O(-2) = Al2O3 +6e
1) Reaction 1 is due to the electric potential that is applied between the anode and cathode.
2) Reactions 2 & 3 describe the growth of the oxide layer.
References:
Self organized formation of hexagonal pore arrays in anodic alumina.
O. Jessensky, F. Muller, U. Gosele., Applied Physics Letters, Vol 72 (10), 1173-1175, 1998
Conditions for fabrication of ideally ordered anodic porous alumina using pre-textured Al. Hidetaka Asoh, Kazuyuki Nishio, Masashi Nakao, Toshiaki Tamamura, Hideki Masuda., Journal of The Electrochemical Society, Vol148 (4), B152-B156, 2001


0 Comments:
Post a Comment
<< Home